AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog

Conversions are given between moles, mass, and molecules. Chemical equation: (N H 4) 2 C r 2 O 7 dissociates into C r 2 O 3, N 2, and H 2 O. 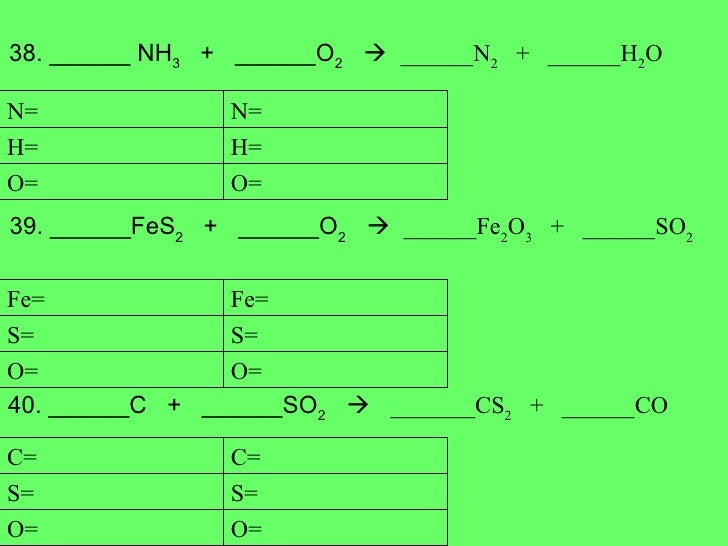
The general equation for a synthesis reaction is: A + B AB. The reactants may be elements or compounds, while the product is always a compound. Balance oxygen (you may need to use a fractional coefficient) If you used a fractional coefficient, multiply by the denominator so all. In balancing carbon based combustion reactions there is a schema that makes things very easy. Because these multi-atom ions generally bond and react as indivisible units, the. CxHyOz + O2 CO2 + H2O (5.3.3) (5.3.3) C x H y O z + O 2 C O 2 + H 2 O. Recall that the nitrate ion, NO 31, is a polyatomic anion. 
On the first row, Total atoms of carbon C (), enter the number of carbon atoms of your substance. Tip 1: When you are trying to balance the chemical equations, you should remember that you can only change the value of coefficient in front of the element or compound, and not the subscript. To use the calculator, enter the molecular formula of your substance. Add a coefficient to the single carbon atom on the right of the equation to balance it with the 3 carbon atoms on the left of the equation. 4: The Relationships among Moles, Masses, and Formula Units of Compounds in the Balanced Chemical Reaction for the Ammonium Dichromate Volcano. A synthesis reaction is a type of chemical reaction in which two or more simple substances combine to form a more complex product. The following reaction is classified as a double replacement because the anionic components of two compounds, the iodide ion, I 1, and the nitrate ion, NO 31, respectively, exchange positions. The combustion reaction calculator will give you the balanced reaction for the combustion of hydrocarbons or C, H, O substances. Use a coefficient to balance the single carbon atom. \): Some Useful Solubility Rules (insoluble) These compounds generally do not dissolve in water (are insoluble):Ĭompounds of Li +, Na +, K +, Rb +, Cs +, and NH 4 +Ĭompounds of Li +, Na +, K +, Rb +, Cs +, NH 4 +, Sr 2 +, and Ba 2 +įor example, consider the possible double-replacement reaction between Na 2SO 4 and SrCl 2. This means that you will need to balance the carbon atoms first.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
